pH
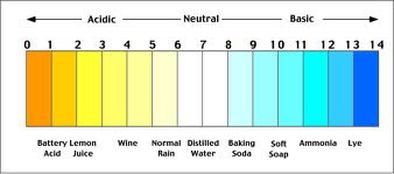
The abbreviated pH means potential Hydrogen or the power of hydrogen. Without getting into a chemistry lesson, it is what we use to measure the alkalinity or acidity of a substance. Clean water should be neutral, which is a 7 on the pH scale. The more acidic a product is, the lower the score will be.
Keep in mind that a food product may be acidic prior to digestion, but alkaline-forming after. This is why a good understanding of proper food combinations is so important.
Keep in mind that a food product may be acidic prior to digestion, but alkaline-forming after. This is why a good understanding of proper food combinations is so important.
A slightly alkaline pH is the key to a balanced body. Our bodies strive to maintain a state of equilibrium when it comes to pH (ideally neutral around 7). However, because most of our foods and drinks are processed and therefore highly acidic, our bodies have to work very hard to constantly neutralize these acids with alkaline minerals such as calcium, potassium, and chloride (salt). Whenever this balance is compromised, dis-ease is the result.
The blood must maintain a pH of 7.35-7.45, with the ideal pH at 7.365. To maintain this balance the body will rob minerals from the bones to buffer acidity. This can lead to osteoporosis and other diseases caused by mineral deficiencies. Often, the very products that are touted as having lots of calcium will actually be the culprit of osteoporosis and other degenerative diseases because the product becomes highly acid-forming in the body and results in more minerals being leeched out of the body to buffer the rising acidity.

Milk is a prime example of a product that is loaded with calcium, but is actually the primary cause of osteoporosis due to its acid-forming nature. Once milk has been processed (undergone homogenization and pasteurization) all the natural enzymes have been destroyed. These enzymes are what deliver the nutrients to your cells and assist your body in digestion. Although milk is fairly neutral on the pH scale, it becomes acid-forming after digestion due to the undigestible protein content. Specifically, the protein casein is very difficult for the human body to digest (keep in mind cows have several stomachs to digest, and the casein is designed to add hundreds of pounds of weight in a short time-frame). Milk also increases the production of mucous (to get rid of the toxic casein protein), which feeds cancer cells. It is better to substitute with rice milk, almond milk, soy milk or goat's milk.

Lemons are another prime example of a food that can be acidic before you eat it and alkaline-forming after digestion. Lemon juice is very acidic at a 2 on the pH scale. However, lemons become alkaline-forming after proper digestion because the overall nutrients in the lemon are alkaline while the citric acid is a fairly weak acid that can be easily neutralized and removed from the body via sweat and respiration, unlike stronger acids that must be eliminated via the kidneys. Acids such as uric acid, which is produced from protein metabolism as seen above in the case of milk, are very strong and must be eliminated through urination. They require more alkaline minerals to buffer the high acidity.
